ICMR Centre for Advanced Research (CAR)

About
About
The Indian Council of Medical Research (ICMR), Government of India has awarded a Centre for Advanced Research (CAR) to the Mother and Child Health department of Interactive Research School for Health Affairs (IRSHA), a constituent unit of Bharati Vidyapeeth University with a grant of over Rs 7.0 Crore.
This ICMR CAR REVAMP (Research Exploring Various Aspects and Mechanisms in Preeclampsia) study, the first of its kind in the country will help in early identification of pregnant women who are at risk of developing preeclampsia. The prospective design of the study for the first time will establish the role of LCPUFA in understanding the underlying biochemical and molecular mechanisms involved in preeclampsia and their association with developmental programming in children.
The study is being carried out at the Interactive Research School for Health Affairs (IRSHA) in collaboration with Bharati Medical College and Hospital, Pune; Gupte Hospital and Research Centre, Pune; and Council of Scientific and Industrial Research-Centre for Cellular and Molecular Biology (CSIR-CCMB), Hyderabad. The current study is ongoing and the total duration of the study is five years i.e. from 31st March 2017 - 30th March 2022.
Introduction
Introduction
Preeclampsia is a major cause of maternal, fetal and neonatal morbidity and mortality, particularly in developing countries. Considering the burden of preeclampsia and its associated complications, it is important to understand the underlying risk factors and mechanisms involved in its etiology. There is considerable interest in the potential for dietary long chain polyunsaturated fatty acids (LCPUFA) as a therapeutic intervention to prevent preeclampsia, as they are involved in angiogenesis, oxidative stress, and inflammatory pathways. The study hypothesizes that LCPUFA and one carbon metabolite status among Indian women play a causal role in the etiology of preeclampsia, mediated by changes in oxidative stress and growth factors which alter placental development. This study proposes to examine the associations of maternal LCPUFA and one carbon micronutrient status in early gestation with clinical outcome in preeclampsia and to understand the underlying key biochemical and molecular mechanisms.
The ICMR Centre for Advanced Research (CAR) study follows a cohort of pregnant women from early pregnancy until delivery to examine longitudinally the associations of maternal LCPUFA with clinical outcome in preeclampsia. A multisite centre for advanced research was established and pregnant women coming to Bharati hospital and Gupte hospital, Pune, India for their first antenatal visit are recruited and followed up at 11-14 weeks, 18-22 weeks, 26-28 weeks, and at delivery. Their personal, obstetric, clinical, and family history are recorded. Anthropometric measures (height, weight), food frequency questionnaire (FFQ), physical activity, socioeconomic status, fetal ultrasonography, and color Doppler measures are recorded at different time points across gestation. Maternal blood at all time points, cord blood, and placenta at delivery are collected, processed and stored at -80°C. The children’s anthropometry is assessed serially up to the age of 2 years, when their neurodevelopmental scores will be assessed.
The Outcome of Proposed Study
The Outcome of Proposed Study
The present study will be useful in:
- Development/validation of biomarkers for early prediction of preeclampsia.
- Providing information on mechanistic aspects of preeclampsia.
- Understanding the neurodevelopmental outcome of children born to women with preeclampsia.
- Correlation of USG and color Doppler findings and histopathological examination of placenta.
- Establishing a biorepository: The establishment of a biorepository will provide an invaluable resource to be shared with other investigators for future research on other factors influencing adverse pregnancy outcomes.
Objectives
Objectives
The REVAMP study proposes to examine the associations of maternal LCPUFA and one carbon micronutrient status in early gestation with clinical outcome in preeclampsia and to understand the underlying key biochemical and molecular mechanisms. It consists of 4 sub-projects:
Project 1: Longitudinal changes in metabolites of the one carbon cycle in preeclampsia
The study will examine baseline levels (in early pregnancy) at 11–14 weeks of gestation (visit 1 –V1) and changes across gestation [18–22 weeks (visit 2 – V2), 26–28 weeks (visit 3 – V3) and at delivery] in components of the one carbon cycle [(folate, vitamin B12, methionine, glutathione, homocysteine, SAM, S-adenosylhomocysteine (SAH)] from early pregnancy until delivery, and placental global DNA methylation levels. It will also examine the relationship of altered one carbon metabolism to methylation potential (SAM: SAH ratio) in the placenta. Furthermore, these changes in the one carbon cycle metabolites will be associated with dietary and lifestyle factors, USG and color Doppler measures.
Project 2: Role of oxidative stress and metabolites of LCPUFA metabolic pathway in preeclampsia
This study will examine levels of oxidative stress markers (protein carbonyl, 8- hydroxyl (OH) guanidine, malondialdehyde (MDA); LCPUFA status and their bioactive metabolites (thromboxane B2; prostaglandin E2) at baseline and across gestation. In addition, measurements of placental phospholipids and gene expression and DNA methylation patterns of PPAR gamma, PEMT, and DNA methyl transferase (DNMT) genes from the placenta will be undertaken.
Project 3: Growth factors and regulation of their gene expression in preeclampsia
This study will analyze levels of angiogenic growth factors – [VEGF, PlGF, sFlt-1, sEng], HIF-1, neurotrophins – [brain derived neurotrophic factor (BDNF), nerve growth factor (NGF) and their receptors] across gestation. It will also examine the associations between altered expression of growth factors involved in placental development, their regulation by methylation as a response to altered one carbon metabolism, and placental histopathological changes. These molecular as well as structural changes in the placenta will further be correlated with clinical parameters for better understanding the etiopathology of preeclampsia.
Project 4: Association of maternal nutrition, biochemical and epigenetic changes with anthropometry and developmental changes in children born to mothers with preeclampsia
This study aims to follow-up the babies for their postnatal anthropometric measurements at birth, 6, 10, and 14 weeks, 6, 9, 12, 15, 18, and 24 months and developmental scales at 2 years of age. The associations of maternal nutrition, biochemical and epigenetic changes, USG and Doppler measures with anthropometry and developmental scores in children will also be undertaken to better understand the mechanisms involved in the fetal programming of developmental disorders in children born to mothers with preeclampsia.
Figure 1 depicts the connectivity between the various objectives.
Key questions
The proposed study will answer the following questions:
- Do levels of LCPUFA and their metabolites, and one carbon nutrients, differ in early pregnancy between women who do and do not go on to develop preeclampsia?
- Is there evidence that these differences are dietary in origin?
- Do the molecular and biochemical analyzes provide a plausible mechanistic pathway to explain the pathophysiology of preeclampsia?
- Do the early pregnancy biomarkers identified provide a clinically useful prediction of later preeclampsia?
- Does the postnatal growth and cognitive development of children of women with preeclampsia differ from that of controls?
- Are these differences in the children explained by changes in growth factors?
Mission
Mission
Reducing maternal and child morbidity and mortality by exploring mechanisms involved in preeclampsia.
Major Activities
Major Activities
Meetings with Collaborators and Advisers

Prof. Caroline Fall (University of Southampton)

Dr. K. Kumaran (MRC Lifecourse Epidemiology Unit & Holdsworth Memorial Hospital, Mysore)

Dr. Giriraj Chandak (CSIR- Centre for Cellular and Molecular Biology (CSIR-CCMB), Hyderabad)
Discussion with Clinicians from Bharati Hospital and Gupte Hospital


Training in Administering Questionnaires

Staff Trained in Administering Nutritional Questionnaires at National Institute of Nutrition (NIN), Hyderabad

Staff Trained in Administering Nutritional Questionnaires at National Institute of Nutrition (NIN), Hyderabad

Staff Trained in Administering Standard of Living Index (SLI) and Physical Activity Questionnaire at IRSHA
Training in Placental Histopathology

Training at St. John’s Research Institute, Bangalore
Staff Counseling the Patients and Administering Various Questionnaires at Bharati Hospital and Gupte Hospital




Follow up Meetings with all the Collaborators

Yearly Advisory Meeting

Establishment of Biorepository



Laboratory Analysis
1. Gas Chromatography (GC)
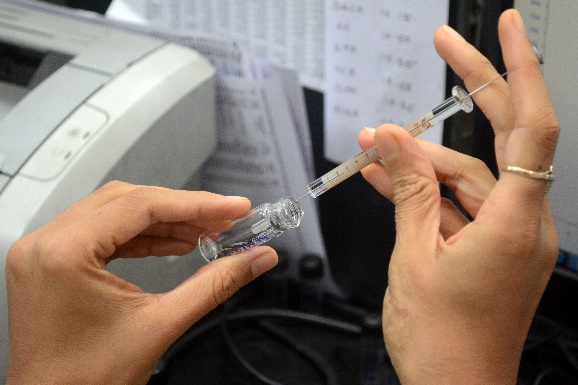

Fatty acid analysis using GC
2. ELISA
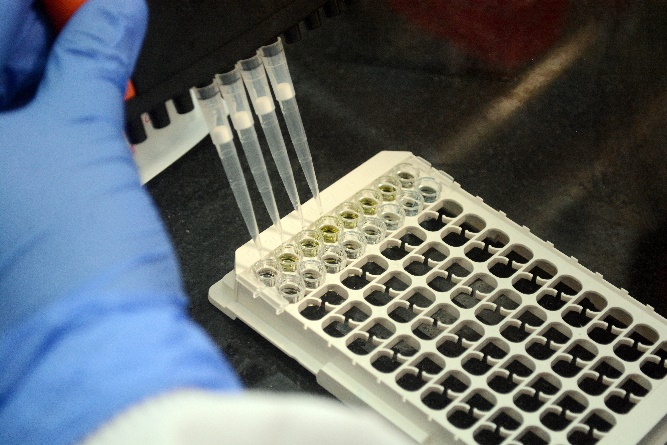
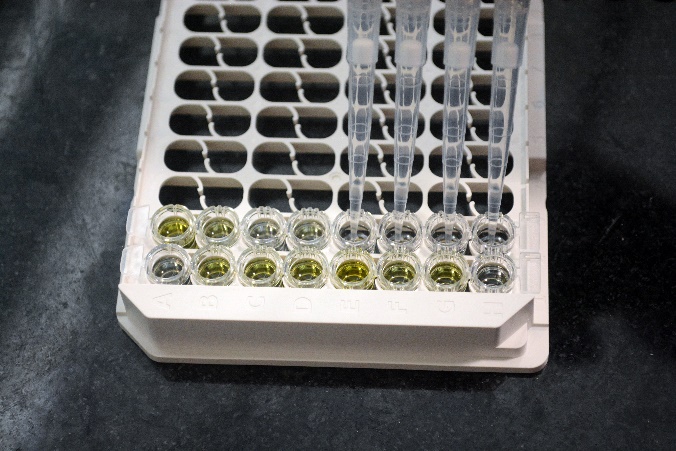
Estimation of protein levels of growth factors using ELISA
3. PCR and Real Time-PCR
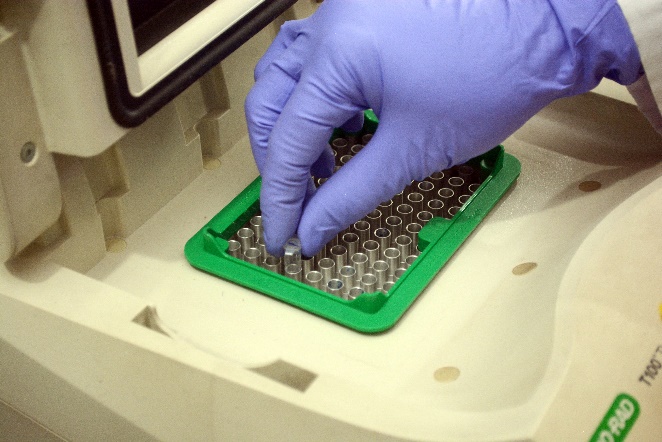


mRNA expression of various genes using RT-PCR
4. High Performance Liquid Chromatography (HPLC)

S-Adenosyl Methionine (SAM) and S-Adenosyl-L-Homocysteine (SAH) estimations using HPLC
5. Ultracentrifuge




Isolation of plasma membranes from placental homogenates
Management team and Staff
Management team
- Dr. Sadhana Joshi, Professor and Head
This email address is being protected from spambots. You need JavaScript enabled to view it. - Dr. Nisha S. Wadhwani, Scientist “B”,
This email address is being protected from spambots. You need JavaScript enabled to view it.
ICMR CAR Staff
- Ms. Madhura Sarda, Psychologist,
This email address is being protected from spambots. You need JavaScript enabled to view it. - Ms. Shweta D Madiwale, Research Assistant,
This email address is being protected from spambots. You need JavaScript enabled to view it. - Ms. Rutuja Tope, Nutritionist,
This email address is being protected from spambots. You need JavaScript enabled to view it. - Ms. Sakshi Selukar, Nutritionist,
This email address is being protected from spambots. You need JavaScript enabled to view it. - Mr. Sagar Bhosale, Social Worker,
This email address is being protected from spambots. You need JavaScript enabled to view it. - Ms. Prachi Joshi, Social Worker,
This email address is being protected from spambots. You need JavaScript enabled to view it. - Ms. Anupam Poddar, Lab Assistant,
This email address is being protected from spambots. You need JavaScript enabled to view it. - Ms. Aditi Mane, Lab Assistant,
This email address is being protected from spambots. You need JavaScript enabled to view it. - Ms. Aditi Godhamgaonkar, Lab Assistant,
This email address is being protected from spambots. You need JavaScript enabled to view it. - Ms. Nikita Joshi, Lab Assistant,
This email address is being protected from spambots. You need JavaScript enabled to view it. - Ms. Juilee Deshpande, Lab Assistant,
This email address is being protected from spambots. You need JavaScript enabled to view it. - Ms. Aboli Ballal, Lab Assistant,
This email address is being protected from spambots. You need JavaScript enabled to view it. - Mr. Mahesh Funde, Data Entry Operator,
This email address is being protected from spambots. You need JavaScript enabled to view it. - Mr. Nganthoi Elangbam, Data Entry Operator,
This email address is being protected from spambots. You need JavaScript enabled to view it. - Mr. Aniket Shelar, Field Attendant,
This email address is being protected from spambots. You need JavaScript enabled to view it.
Contact us
ICMR- Centre for Advanced Research (CAR)
Interactive Research School for Health Affairs (IRSHA),
Bharati Vidyapeeth (Deemed to be University),
Pune-Satara Road, Dhankawadi,
Pune 4110043,
Maharashtra, India
Tel No.:- 020-24366920
Emails :- This email address is being protected from spambots. You need JavaScript enabled to view it. , This email address is being protected from spambots. You need JavaScript enabled to view it.



